Regulatory Requirements for Biodistribution Studies for Gene Therapy Products
As we know, these mRNA products were assessed as ordinary vaccines and not as gene therapy products. However, even for gene therapy products, the regulatory requirements for biodistribution studies are still product specific and tailored to each RNA therapeutic. Moderna escaped the requirement for biodistribution studies, relying on its one IM biodistribution study of its failed CMV vaccine discussed in The Biodistribution of Lipids, Part 1. Pfizer performed one very confusing and poor biodistribution study which was not accepted, so the second biodistribution study was performed starting in July which was well into the start of the human clinical trials! This one is colloquially known as the Japanese study but every regulator got the same information by Nov 2020.
What needs to be measured?
Overall, depending on the construct of the RNA therapeutic, the preclinical biodistribution studies may include:
the carrier-delivery system (ie the LNP specifically)
individual components of the delivery system (ie the mRNA AND the individual lipids. especially the novel lipids)
the expressed spike protein
the combined final product (ie the LNP/mRNA and biocorona particle)
This is not easy. It is time consuming and requires expertise and cannot be done in vivo in humans.
How long to measure biodistribution?
This depends of course on the dose, the carrier, the route of administration and the expressed protein but generally should continue until the signal becomes undetectable or until a long-term plateau phase is reached. So that is not just 48hrs or even 7 days; for the mRNA vaccines that might mean upto 60 days or more depending on how long that spike protein is expressed.
Plus the right animal model should be used. Not only do you want the animal to translate the spike protein similar to humans, they should have the same immune response to the spike protein. This makes rats perhaps a poor animal model and even non human primates may not be comparable enough. And its time consuming and costly. This is why the Bitounis article discussed iv vitro and ex vivo alternative models to animal testing. This is an active research field but demonstrates how difficult it is to measure and test these multicomponent nanoparticle products.
Tissue panels that should be studied
Ohhh this is important. What tissues should you look at? This is an evolving field, and the EMA is much stricter than the FDA. Just for fun, I compared what the EMA requires for biodistribution of gene therapy products compared to the tissues examined by the pivotal Pfizer distribution study.
For the sake of brevity, here are the tissue panels the EMA would request (which includes repeated dosing) that were INCLUDED and those NOT INCLUDED in the Pfizer biodistribution data of the tissues they examined
INCLUDED
Adipose tissue, brain and spinal cord, heart, adrenal glands, bladder, bone (femur), bone marrow, eyes, injection site, kidneys, stomach and large intestine, liver, lung, lymph nodes (mesenteric and mandibular), ovaries and uterus, pancreas, pituitary gland, prostate, salivary glands, skin, small intestine, testes, thymus and thyroid.
NOT INCLUDED in PFIZERS BIODISTRIBUTION STUDY
BLOOD SMEARS, lung bronchioles and bronchi, AORTA, OPTIC NERVES, larynx, MAMMARY GLANDS, esophagus, PERIPHERAL NERVES, skeletal muscle, tongue, parathyroid glands, subcutaneous tissue, ureters and vagina.
A few of the excluded tissues are relevant to potential vaccine damage, specifically the aorta, blood, peripheral and optic nerves and mammary glands for breastfeeding.
Techniques used
QWBA (quantitative whole body radiography). First, a radioactive isotope is incorporated in the investigated compound. So radioactive carbon for the cholesterol in the LNPs was used in the Pfizer study for example. A major
limitation is that the expressed protein (spike protein) is not tracked
and the this technique cannot discriminate metabolites and degradation
products of the LNPs.
FISH (fluorescence in situ hypbridization) is the gold standard for a single RNA molecule in fixed samples and has been used for mRNA detection. This technique could also target multiple mRNAs like for the bivalent vaccines. You might also want to use PCR or ddPCR to detect small quantities of mRNA
LC-MS/MS can be used for the synthetic lipids (individually) for example to differential from QWBA and degredation products
all kinds of fluorescent probes
It is assumed that changing the coding sequence of the mRNA is unlikely to have a significant impact on the physiochemical properties and distribution of the mRNA/LNP, then one can substitute luciferase mRNA to monitor distribution. This was used as justification for Pfizer and Moderna in their biodistribution studies. I believe that assumption should be verified, as how the mRNA packs within the LNP can affect size and shape and biocorona. Might not be that much though.
Also not ALL mRNA GETS INTO THE CYTOSOL. So you could have mRNA floating around in the lymph or blood and feces and somehow you need to separate that mRNA from the small amounts that get in the cell.
An Example of a Complete Biodistribution Study
Well Moderna just did a very thorough biodistribution study of its Lipid 5, which is very similar but not the same (of course!) as SM-102 which was used in the vaccine trials. In this study, they using a modRNA of Factor IX (N1-methylpseudouridine-substituted mRNA was synthesized in vitro from a linearized DNA template).
The actually synthesized LNPs which were radiolabelled and assessed by QWBA containing the Factor IX mRNA (nontranslating) and LNPs which were not radiolabelled (analyzed via LC-MS/MS) and contained the same size of mRNA, but this mRNA was not specified. (grrr)
The main purpose was to see how the intact LNPs are biodistributed and then the individual component parts.
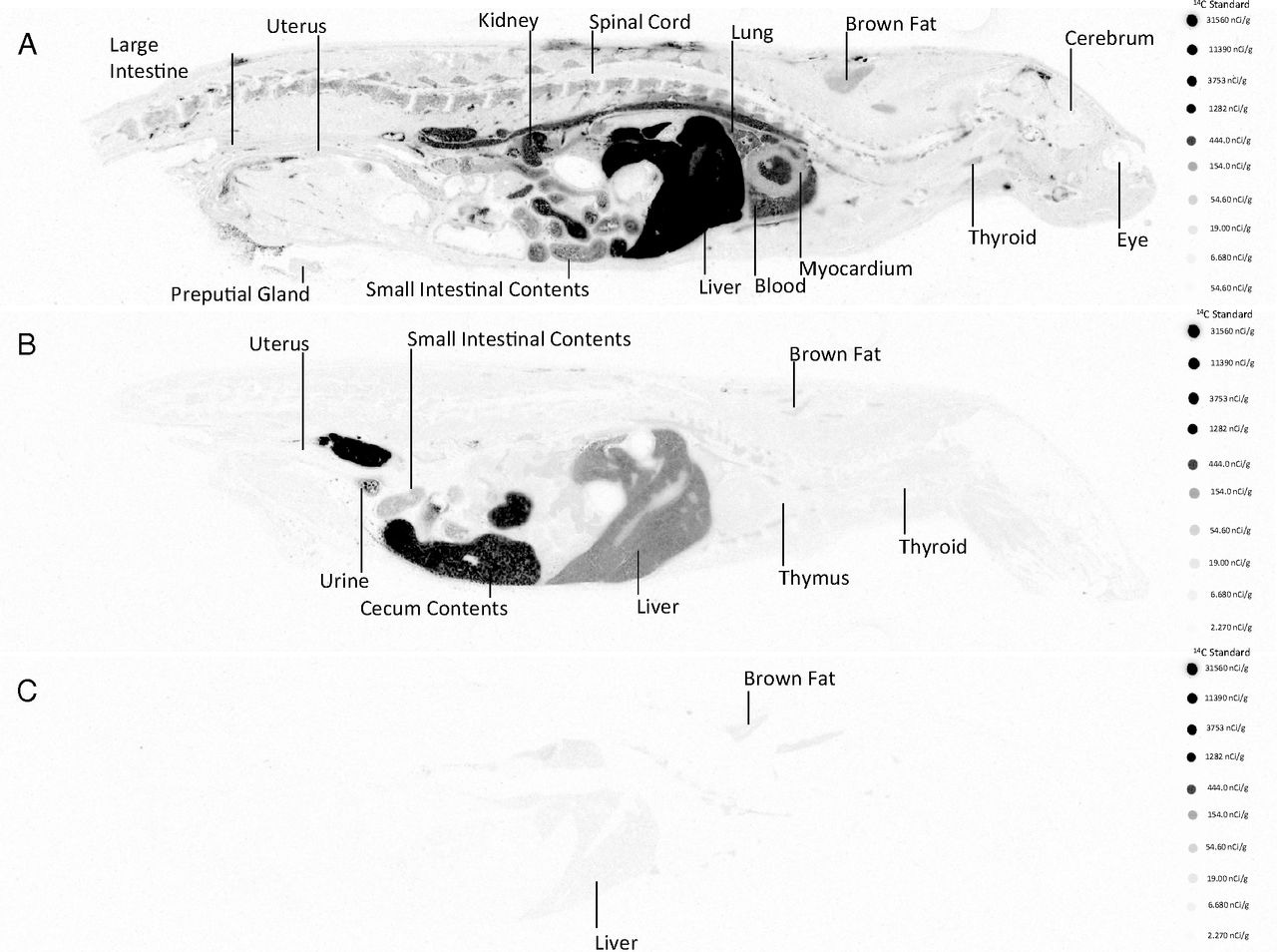
Here is the QWBA data
Representative whole-body autoradiographic slices displaying radioactivity measured after intravenous administration of [14C]Lipid 5-containing LNPs in female Sprague Dawley rats. A single dose of [14C]Lipid 5-containing LNP encapsulating nontranslating factor IX (NT-FIX) mRNA was administered at 2 mg/kg of encapsulated mRNA via 10-minute intravenous infusion. Representative slices shown of slice 4 of whole-body sections at (A) 1 hour postdose, (B) 10 hours postdose, and (C) 168 hours postdose
As expected after IV administration, most of the lipid goes to the liver but Holy Toledo, look how much is in the lung, heart and kidney at one hour, which then is all accumulated in the cecum and large intestine contents at 10hrs. It is mostly gone at 7 days, though some is still in the brown fat and liver.
Here is a better look over time at the radioactivity of basically the intact LNP/mRNA particles
Mean detectable radioactivity in blood and tissues in Sprague Dawley rats (1 male and 1 female) as measured by quantitative whole-body autoradiography. (A) circulatory system, (B) digestive system, (c) urinary system + adrenal gland, (d) musculoskeletal system, (e) nervous system, (f) reproductive system, (g) immune system, (h) respiratory system. Int, intestinal.
Well here we see biodistribution similar to the Pfizer study; liver, spleen, adrenals and ovaries are still showing radioactivity at 7 days and appear to plateau. Notice the intact LNPs are gone from the heart in <24 hrs.
Next are UNLABELLED LNPs measured by LC-MS/MS
Mean (±S.D.) detectable plasma and tissue Lipid 5 concentrations after intravenous administration of Lipid 5-containing LNPs in Sprague Dawley rats as measured by LC-MS/MS. a) circulatory system, b) digestive system, c) urinary system, d) muscoskeletal system, e) nervous system, f) reproductive system, g) immune system, h) respiratory system.
These are more representative of broken up LNPs and the individual lipids. Although similar there is significant individual lipids in the plasma and heart, jejunum and even the lung. Guess the lung doesn’t deal with intact LNPs too well as expected considering the lung barrier and other issues with size and biocorona of LNPs. Maybe people are breathing out LNP components? Unlikely intact LNPs. BUT. What we see in these 3 images is enterohepatic circulation of the LNPs and their component parts which are excreted via the feces. Does this have implications for transfection of the microbiome? I wonder if that is also happening with the vaccines and I expect so. LNP components are also being excreted whole and degraded through the urine. No data reported on the ovaries, just the uterus.
The mRNA
OK now the good part. Where is the mRNA found?
Mean (±S.D.) detectable plasma and tissue mRNA concentrations after intravenous administration of Lipid 5-containing LNPs in Sprague Dawley rats. a) circulatory system, b) digestive system, c) urinary system, d) muscoskeletal system, e) nervous system, f) reproductive system, g) immune system, h) respiratory system. BMF, bone marrow femur.
WELL WELL WELL. Here we have the heart exposed to mRNA for 48 hours, and so too the plasma. Is it in the macrophages? RBCs? or just naked floating around from the broken up LNPs? The kidney and skeletal muscle have lots of mRNA but muscle didnt have much LNPs in the previous figures. What gives? And lookie here at the EYES. LOTS of mRNA in the eyes but little LNPs and individual broken up LNPs. So I am hypothesizing only a little bit of LNPs get into the eyes but it looks like all of the mRNA gets released or all the LNPs that go to the eye get transfected. Here is an example where LNPs biodistribution does not equal mRNA distribution. Also the lung is trying hard to get rid of the mRNA but it is sticking around even past 7 days. Is it naked mRNA in the lungs? And a expected lots of mRNA is found in spleen, lymph nodes and thymus.
Finally, protein production. HOLY TOLEDO
Mean (±S.D.) detectable tissue intracellular protein concentrations after intravenous administration of Lipid 5-containing LNPs in Sprague Dawley rats. a) circulatory system, b) digestive system, c) urinary system, d) muscoskeletal system, e) nervous system, f) reproductive system, g) immune system, h) respiratory system.
What do we see here? The heart is pumping out protein for 7 days despite the previous graph which suggests the mRNA is minimal by then. OR maybe it plateaus off and they didnt graph it as such. Remember the heart gets a big load of the LNPs for a few hours which then dissipates. But we get protein production that appears to plateau. This may explain a lot. I expect the liver to keep making protein, but the jejunum???? What does that signify? Anyone? Thank goodness the liver has a huge capacity for detoxification and renewal. Whew. And the kidneys are more affected than we think. Maybe we should be looking at kidney impairment as caused by the vaccine. These products are hard on our excretory organs, the liver, the kidneys and you can also see here, the lungs. We don’t have any data on skin biodistribution, mRNA or protein production but my guess is the body is trying to get rid of the product via the skin. Skeletal muscle is not affected so even they may have lots of mRNA floating around it is not being translated in the skeletal muscle. This is a tissue which does not appear to be transfected easily.
Ovaries are only measured for 72hrs but it sure looks like there is a LOT of protein production continuing in the ovaries despite not that much mRNA. And look at the poor thymus. This is not good. Especially for children.
Here is the pdf with supplementary data of this interesting study so you can look at this in more detail and see what you think these results mean.
Biodistribution of Lipid 5, mRNA and its translated protein
Here is what Moderna says about the biodistribution of its lipid 5.
The results from this study demonstrate that Lipid 5-containing LNPs are distributed throughout tissues after initial intravenous administration and Lipid 5 is rapidly eliminated thereafter. The time course and radio-activity distribution patterns of [14C]Lipid 5-containing LNPs, suggest that Lipid 5 and its [14C]metabolites undergo elimination by both biliary and renal routes with no indication of redistribution or prolonged metabolite exposure.
No mention of tissue mRNA levels and prolonged protein expression by the mRNA in this construct. The mRNA used N-1-methylpseudouridine but I cant tell if it is further codon optimized. But hey, this lipid is more biodegradable! Isn’t that just great?/sarc
Summary
the regulatory requirements for a thorough biodistribution assessment of LNP/mRNA products is evolving
they are difficult and time consuming to do and pharma is looking at ways to streamline and eliminate costly steps
in vivo animal models (usually rats) are currently being used but this may not be appropriate. Other types of models are required but none of these studies can be done in humans. Even in rodents these studies are pretty yucky.
At least in LNPs using lipid 5, the intact LNPs do not last long in the plasma and are broken up quickly and excreted through normal urinary and biliary channels. I also suspect the lung and skin are used. I also suspect similar issues are happening with the vaccine.
The relationship between intact LNP/mRNA particles and mRNA transfection is comparable but can differ in several tissues.
The relationship between mRNA amounts and protein production are not always correlated depending on the tissue and/or organ.
It appears all LNP/mRNA particles have distribution to ovaries, adrenals, spleen and liver despite the molecular chemists trying to come up with better and “safer” synthetic lipids. This is likely because the LNPs are particles that look like cholesterol and go through fenestrated epithelium easily. Where can fenestrated epithelium be found? Yup, ovaries, testes, kidneys, endocrine glands like adrenals, pancreas and thyroid. Oh and the small intestine like the jejunum. Sound familiar?
The LNPs don’t distribute everywhere throughout the body and certainly do not distribute evenly as we expect with small molecule drugs.
FINALLY WE NEED TO TALK ABOUT EXCRETION
This is when we will talk about exosomes. All that naked mRNA floating around? All the bits of broken down LNPs? They are likely in exosomes floating in the plasma and extracellular fluid. These exosomes will be discussed in Part 3 of the biodistribution of the lipids.
I am still learning exactly what happens to these LNP/mRNA particles and its associated toxicities. There is still a lot to parse in this particular Moderna lipid study and I would appreciate any insights.
Thanks for reading.
Rant over.
Pray the rosary







The best way to describe LNPs to anyone is "A razor edge that cuts through any cell skin" and people understand pretty fast.
Also when you look back into LNPs and their design process you see they were created, specifically, to pierce through membranes, since this was a "cancer treatment" at first. And still is.
I just watched this (thanks to https://genervter.substack.com/p/die-phospholipide-doppelschicht-und?publication_id=1003447&post_id=141790580&isFreemail=true&r=22x7dz&open=false)