Could Residual DNA Be Functional Rather Than A Contaminant or Impurity?
A speculative exploration of an overlooked manufacturing consideration
Introduction
As many may know, I have been working peripherally on the residual DNA levels since 2021 when I started reading the EMA EPARs and then the leak/hack. Mostly regarding the regulatory aspects, the batches and the manufacturing process. But for the last 3 years or so I have been focusing on the LNPs. When Genervter Bürger posted this:
Having said that, and keeping the authors’ observation regarding RNA integrity in mind, what would be the obvious logical question now?: Was DNA really an “accident” or a sixth particle?
something clicked. AHA! I thought. There might be something to this.
And the followup post by Genervter Bürger talked about cholesterol crystals
The authors generated DNA-cholesterol adducts via covalent coupling and combined these with single-stranded DNA overhangs to physically prevent the uncontrolled clustering of the cholesterol units. In doing so, they observed an interesting effect: the overhangs forced the cholesterols into controlled clusters of four, and the expected formation of a crystal lattice was suppressed.
Now this is similar chemically on how alloys work and become more stable.
And he also makes these observations
As early as the 1970s and 80s, it was established that SV40 transformation specifically alters cell membrane architecture and cholesterol ester accumulation (Lago et al., 1977; Chen et al., 1988).As early as 1980, Fraley and Papahadjopoulos–pioneers of lipid-based delivery–demonstrated the successful encapsidation of SV40 DNA within liposomes to facilitate cellular entry, proving that DNA and lipids form a functional, albeit complex, unit.Crucially, the SV40 large T antigen has been shown to rely on tightly associated lipids as anchors within the plasma membrane (Henning et al., 1983). When we look at modern findings on the rapid, two-state mechanism of RNA encapsidation by SV40-derived nanoparticles (Kler et al., 2012)a clear pattern emerges: the virus uses a biophysical “blueprint” to organize lipids. In the LNP context, DNA fragments may be inadvertently replicating this viral strategy: not as a genetic message, but as a structural anchor. 4 , 5, 6, 7, 8
Hmmmm.
Can nucleic acid fragments alter structural degrees of freedom within an extremely densely packed LNP system? This is a question of materials science, not virology. This hypothesis is scientifically valid, even if it currently remains unproven
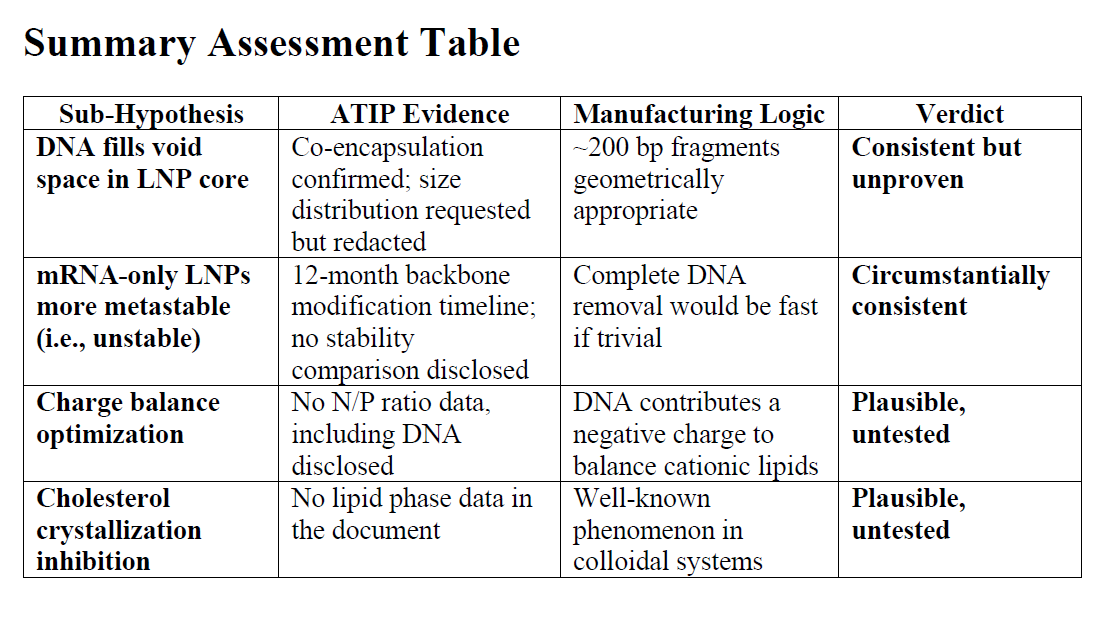
Now this is purely a speculative hypothesis, a thought experiment at this time, but it is grounded in LNP chemistry and physics we reviewed, and on what I know of the manufacturing process. Plus strong circumstantial evidence from a Health Canada ATIP (ie FOIA)
The Core Question
What if the residual plasmid DNA in mRNA vaccines isn’t just an process-related impurity to be minimized, but rather a functional component that manufacturers learned (or knew) contributes to lipid nanoparticle stability? What if?
The Hypothesis in Plain Language
What is the standard narrative?
We usually assume the residual DNA from the bacterial manufacturing process is an unavoidable contaminant that should be reduced to the lowest possible levels for safety reasons.
Here is my alternative hypothesis
The DNA fragments remaining in the final product may actually have a functional purpose and serve multiple stabilization functions:
Structural scaffolding within the LNP matrix
Charge balance optimization between nucleic acids and cationic lipids
Cholesterol crystallization impairment that maintains LNP fluidity
Rather than DNA being purely “added contamination,” it might be “deliberately not removed beyond a certain threshold” because complete removal could compromise product stability.
Why This Hypothesis Might Have Legs: Manufacturing Perspective
The LNP Stability Challenge
Lipid nanoparticles are inherently unstable systems. They face:
Lipid aggregation
Cholesterol crystallization
Charge imbalances
Structural degradation during storage
One of the first papers I read in 2021 was the classic Schoenmaker paper.1 It is even more relevant today.
What DNA Fragments Could Provide
1. Void Space Filling (Molecular Packing Peanuts)
mRNA alone may not efficiently fill the LNP core
DNA fragments (~200 bp range) could fill gaps between mRNA molecules
This could reduce internal mobility and prevent structural collapse
Similar to how packing peanuts prevent cargo from shifting during transport
2. Charge Balance Optimization (N/P Ratio Fine-Tuning)
Cationic lipids need negatively charged nucleic acid partners
The ratio of total negative charges (mRNA + DNA) to positive charges (lipids) might be optimized by DNA presence
Pure mRNA alone may not provide ideal charge distribution throughout the particle, mixed polyanions are better
DNA could contribute to the overall electrostatic environment
3. Cholesterol Crystallization Impairment
DNA fragments could interfere with cholesterol crystallization patterns
This would maintain membrane fluidity critical for cellular uptake
Prevents phase separation during cold storage
Similar to how impurities in chemistry prevent unwanted crystallization. This is my favorite subhypothesis.
4. Assembly path modification
that is how the LNP is formed around a nidus of mRNA, mixed nucleic acids of polyanions make it easier for the condensation of the LNP
Biocorona modification
If DNA is on the outside of the LNP this could affect the biocorona and targeting as briefly discussed below
The Regulatory Evidence: What Health Canada Documents Reveal
So what really got my almonds going was the ATIP HC-2004-0097 filed by Scoops McGoo in his substack post below.
but I have also put it up on Research Gate.
This is the 56-page report by Pfizer in response to Health Canada’s questions regarding the residual DNA levels and the SV 40. We have looked at this before.
Following questioning on findings of undisclosed DNA in Covid vials, Health Canada required Pfizer to produce a safety report on the newfound vaccine contamination — due date: December 1, 2023. The existence of the request by Health Canada for such analysis was discovered in government internal emails.
The Smoking Gun: Co-Encapsulation Confirmation
In Health Canada ATIP document A-2024-000097, Pfizer explicitly states:
“Residual DNA encapsulated within the LNP would be delivered to the cytoplasm with the vaccine mRNA.”
This is critical. Pfizer confirms that DNA fragments are physically inside the LNPs, co-localized with mRNA. Not just floating free in the solution, but geometrically positioned within the nanoparticle structure where they could:
Fill void space
Contribute to internal charge balance
Affect lipid packing arrangements
This is not speculation. This is regulatory fact. Though we all suspected this, but the language used is deliberate, imho. It is not accidentally delivered or that it is present and they dont know where it goes, but instead that it is known to be delivered to the cytoplasm. There is ALSO DNA outside the LNPs. Are they attached to the LNPs? What? Another important questions, because researchers are making crosslinked LNPs with cholesterol for stability. In fact, this paper states
Further engineering involved additional DNA coating and external envelopment by plasma proteins. This strategy was based on prior findings demonstrating the peculiar ability of DNA to reverse the charge of cationic lipids16 and facilitate the adsorption of an opsonin-deficient protein corona17.
So if residual DNA is decorating or coating the LNPs, then the protein corona is affected and less likely to be opsonized. What are the implications?
The Size Distribution Question
Health Canada specifically requested:
“Additional data and/or information characterizing the size distribution of residual DNA fragments and residual intact circular plasmid.”
This is likely in response to Kevin McKernan’s presentation to VRBAC (pg 114-) that started this whole thing, and the FDA guidelines to have residual DNA fragments to be <200bp.
However, the ICH Q6B guidelines for residual DNA limits (measured in ng/dose) are size-agnostic. They don’t care if you have 10 ng of 50 bp fragments or 10 ng of 500 bp fragments, the safety limit is purely about total quantity. For the EMA and HC, I think this is what they used.
The reason fragment size matters for my hypothesis is if it affects:
How DNA packs within LNPs
Structural properties of the formulation
Stability characteristics
The ~200 bp fragment size range happens to be precisely the scale that would fit compactly within LNP void spaces between mRNA molecules.
The 12-Month Timeline: Seriously?
Pfizer acknowledged they are evaluating plasmid backbone modification to remove non-essential sequence elements, with preliminary results expected 12 months after correspondence.
Let that sink in. Twelve months to remove sequence elements from a plasmid.
As Kevin McKernan has said in the past, for molecular biologists: that’s an extraordinarily long timeline. Standard plasmid modification can be accomplished in weeks. Removing a sequence element, re-validating the construct, and running a production batch should take months at most.
Why would this take a year?
Two possibilities:
Scenario A: Technical Complexity
Removing elements affects plasmid yield
Changes linearization efficiency
Alters in vitro transcription (IVT) performance
Affects downstream DNA fragment distribution
Any of these would indirectly affect residual DNA content and fragment size patterns
Scenario B: Functional Studies Required
Pfizer is conducting studies to confirm removal doesn’t affect Drug Substance quality attributes
This would only be necessary if there’s concern that DNA levels or composition affect LNP performance
You don’t spend 12 months proving something is irrelevant
Either way, the timeline suggests complexity inconsistent with “trivial inert residual impurity.”
Health Canada’s Extraordinary Statement
This is the most legally and scientifically significant passage in the entire document:
“Based on current scientific knowledge, it is highly unlikely that the elements under discussion could serve a purpose and may be needed to maintain structural integrity... Health Canada remains open to reconsider, should data supporting an impact on plasmid DNA/DS manufacturing process or DS quality attributes be presented.”
Let’s unpack this carefully:
“Highly Unlikely” ≠ Impossible
This qualifier acknowledges the possibility exists. In regulatory language, “highly unlikely” with an invitation to reconsider is remarkable restraint but not dismissal.
“Structural Integrity”
Health Canada is discussing structural integrity of the plasmid template, but the conceptual foundation is identical: nucleic acid architecture affects formulation structure. This language was always a question to me, since it did not make sense.
“DS Quality Attributes”
What in the world would residual DNA have to do with the mRNA CQAs? This is HC opening the door to Pfizer.
Drug Substance (DS) quality attributes (CQAs) include:
LNP particle size and distribution
Polydispersity index (PDI)
Encapsulation efficiency
Colloidal stability
Storage stability
Health Canada has explicitly stated they will reconsider if data shows DNA affects these parameters.
This is essentially the experimental brief for testing the “molecular packing peanuts” or function of the DNA hypothesis, asked by the regulator themselves.
The Specification Asymmetry
The acceptance criterion is:
Upper limit: ≤ 330 ng DNA/mg RNA or < 10 ng/dose
Lower limit: None specified
If residual DNA were purely a contaminant to minimize, you would expect:
Specifications like “as low as reasonably achievable”
Continuous pressure to drive toward zero
Lower limits to ensure consistency
The absence of any lower limit is consistent with the hypothesis that a functional floor may exist, though it doesn’t prove it. In usual manufacturing (and for other contaminants like dsRNA) in other pharmaceutical processes, these typically have “Not More Than [limit]” (i.e. NMT) with aggressive reduction targets. I saw this wording for dsRNA. But NOT residual DNA.
The Half-Life Detail: Mechanistic Relevance
Pfizer states:
“In the cytoplasm, residual DNA is not transcriptionally active and is rapidly degraded with a half-life of 50-90 minutes.”
This is offered as safety reassurance, but consider the timeline:
If DNA is degraded in the cytoplasm after endosomal escape, it was physically present within the LNP during
Manufacturing
Purification
Fill/finish
Storage (weeks to months at -80°C to -20°C)
Thawing
Injection
Circulation
Cellular uptake
Endosomal escape
Any structural role would be operative throughout the LNP’s entire functional lifetime until cytoplasmic delivery, which is exactly when you’d want that stability. So it is not degrading in storage but only once delivered in the cytoplasm as per Pfizer. And they were very specific at mentioning this. And it was not redacted. HMMMM.
The Redaction Pattern: What is Redacted Specifically
The most scientifically significant aspect of this document is what is not there.
The following data, which would directly test the hypothesis, is either absent or redacted:
Missing Data
DNA encapsulation efficiency (% inside vs. outside LNPs). This would tell us how much was thought to be needed for structure
DNA:mRNA co-localization ratio per particle. How many DNA fragments per LNP?
Fragment size distribution requested but redacted. The ENTIRE response is redacted. I have always thought that was very weird, it is a straightforward question. SOME of this is not controversial.
LNP size or PDI vs DNA content correlation. Does the DNA affect colloidal stability?
N/P ratio calculations including that of DNA contribution. This is a CQA, Does DNA contribute to charge balance?
Lipid composition and cholesterol content. Is cholesterol at crystallization risk concentrations? Some data support this.
Batch data tables all redacted
Health Canada’s specific position on DNA limits. Is there an implied functional floor?
This hypothesis doesn’t require conspiracy per se, just industrial optimization. Companies regularly work within manufacturing constraints, and what starts as a limitation can become incorporated into the process design.
Question to ponder: If you were formulating a stability-challenged product at massive scale, would you optimize within your real-world constraints, or chase theoretical purity at the cost of product performance? You know the answer to that.
For Scientists
This is testable. The experiments are straightforward:
Systematic DNA titration studies with stability endpoints
Structural characterization of LNPs across DNA content ranges
Cholesterol crystallization studies in the presence/absence of DNA fragments
The ask: Consider that regulatory specifications may reflect functional optima rather than just safety margins.
But Wait! There is MORE!
What if the residual DNA isn’t cleaned up as much as it should be, because it is in there for an ADJUVANT EFFECT?
I floated this theory in my very first substack
Then this paper by Siguna Mueller, Ph.D., Ph.D. hints at this.
And of course, an exploration and explanation by Anandamide (Kevin McKernan) is various substack posts. This most recent one talks about RNA:DNA hybrids
Pfizer wants the RNA:DNA hybrids in the shots but they don’t want the regulators to know its there. Why would Pfizer want these additional inflammatory molecules in their shots? Do they not illicit the desired response without them?
Are they providing adjuvant-like effects? But could the RNA:DNA hybrids also provide stability and other benefits as I discussed earlier AS WELL?
I put this question to Claude re the RNA:DNA hybrids. Here’s what it said.
Hybridization Solves Problems The Others Don’t
Problem: How does LNP maintain stability for months at -20°C?
Structural alone: Plausible but mRNA should still degrade
Adjuvant alone: Doesn’t explain storage stability
Hybridization: Direct RNase protection mechanism ✅
Problem: Why does fragment size matter so much?
Structural alone: Packing efficiency (weak explanation)
Adjuvant alone: CpG density (possible but incomplete)
Hybridization: Optimal length for stable base-pairing ✅✅
Problem: Why 12 months to modify plasmid?
Structural alone: Some complexity
Adjuvant alone: Need to maintain CpG content
Hybridization: Must maintain complementarity to mRNA ✅✅✅
Problem: Why hasn’t DNA been removed?
Structural alone: Manufacturing constraint
Adjuvant alone: Would reduce efficacy
Hybridization: Product would be completely unstable ✅✅✅
Holy Toledo!!!
If True We Then Have a Triple Functions For the Residual DNA
DNA is not just beneficial , it may be essential
Complete removal could render product unstable/ineffective
Specifications reflect optimized triple function (adjuvant, stability, hybridization)
“Residual impurity” framing might be misleading
What we have here is functionally a quaternary formulation component (lipids + mRNA + DNA hybrids + DNA)
GULP!
Is There ENOUGH DNA?
The mass of DNA is not a lot really when compared to the mass of the mRNA in these LNPs. As Kevin McKernan pointed out to me it is likely 1:30->1:100 ratio. Is that enough to act as a scaffold? This assumes that the DNA acts stoichimetrically. But that is not what is needed. I asked SciSpace to provide an estimate of how much DNA is required for the 5 subhypothesis delineated earlier (charge complementarity, steric void, assembly path modifications, anti-nucleation for cholesterol and biocorona modification) and it provided the following:
How much DNA is required implicitly assumes that DNA must act stoichiometrically — like a reagent that must be present in equimolar quantities to its target. This is incorrect for all four proposed mechanisms. Polyelectrolytes inhibit crystal nucleation at sub-percent surface coverage. Charge perturbations of 10% alter mesophase geometry. Three DNA molecules per LNP are sufficient to fill the void space. The Donnan effect transmits internal charge to the surface at measurable levels. Steric void filling — the most conservative and model-independent calculation — shows that 3,000 ng/dose is 1.6× the required mass for complete void occupancy. The objection is not supported by biophysical first-principles.
I have all of its calculations as well. Sobering. Dont understand a word, BTW. But again, this is just based on first principles, and not on empirical knowledge.
Closing Thoughts
This is speculative. Maybe wrong. But it’s grounded in real manufacturing considerations.
The lipid nanoparticle is a complex, metastable system. DNA fragments ESPECIALLY the RNA:DNA hybrids with their unique charge, size, and structural properties could plausibly contribute to that system’s functionality. Whether this was engineered, discovered, or is entirely wrong is an open question.
What’s needed: Transparency about formulation optimization studies. Data on stability/functionality vs. DNA content. Honest conversation about what “residual” means when it might be functional.
What’s not helpful: Dismissing the question without investigation, or assuming malice when optimization might explain the observations.
What are your thoughts? What evidence would move your belief up or down on this hypothesis?
Disclaimer: This is a speculative hypothesis for discussion purposes. It does not constitute medical advice, claims of wrongdoing, or definitive conclusions about vaccine formulation. All mRNA vaccines have been approved by regulatory authorities and found to be safe and effective by those standards. HAHAHA. Take your electrolytes.
As always continue to pray the rosary.
I have a very heavy substack next week and will be busy with my family for the foreseeable future. See you on the other side.
https://pmc.ncbi.nlm.nih.gov/articles/PMC8032477/pdf/main.pdf




Yes, tragic. Yes, I have long though that DNAs play a role in LNP composition and function. As for them playing a role in immunity - the work you know about and which I had essentially finished in June of 2025, is under review AGAIN. This is now the THIRD major revision - the first was submitted ages ago, then underwent peer-review, and, after I had rewritten some 80-90% of the original version, was fully accepted..... Then, the nightmare unfolded. I am not revealing details. Suffice it to say, what unfolded was worse than I ever thought could happen. After many months of "nightmare," I rewrote it and resubmitted it again. Meanwhile, I am writing a book about related immune issues and have been busy enough and actually appreciate the fact that I have not heard back in a while...
I am envious that you people can throw ideas and research work at each other. I dearly wish I had the education or brains to understand most of what is being said. As a shot injured person, I wonder why testing the subjects isn’t involved to assist in finding answers. Why haven’t diagnostic tests come to market? Judging from what I’ve read, HC seems to have an inkling that all is not well with these shots. Also, I’d love to know why some people I know have had 11 or 12 shots with no side effects and I’ve had one which is a continuing nightmare. Do these shots affect people who share some genetic pool? Thank you for the work that you all are doing. What would we do without good people?